Tiny
implant
OZURDEX® TARGETS MULTIPLE
INFLAMMATORY CYTOKINES
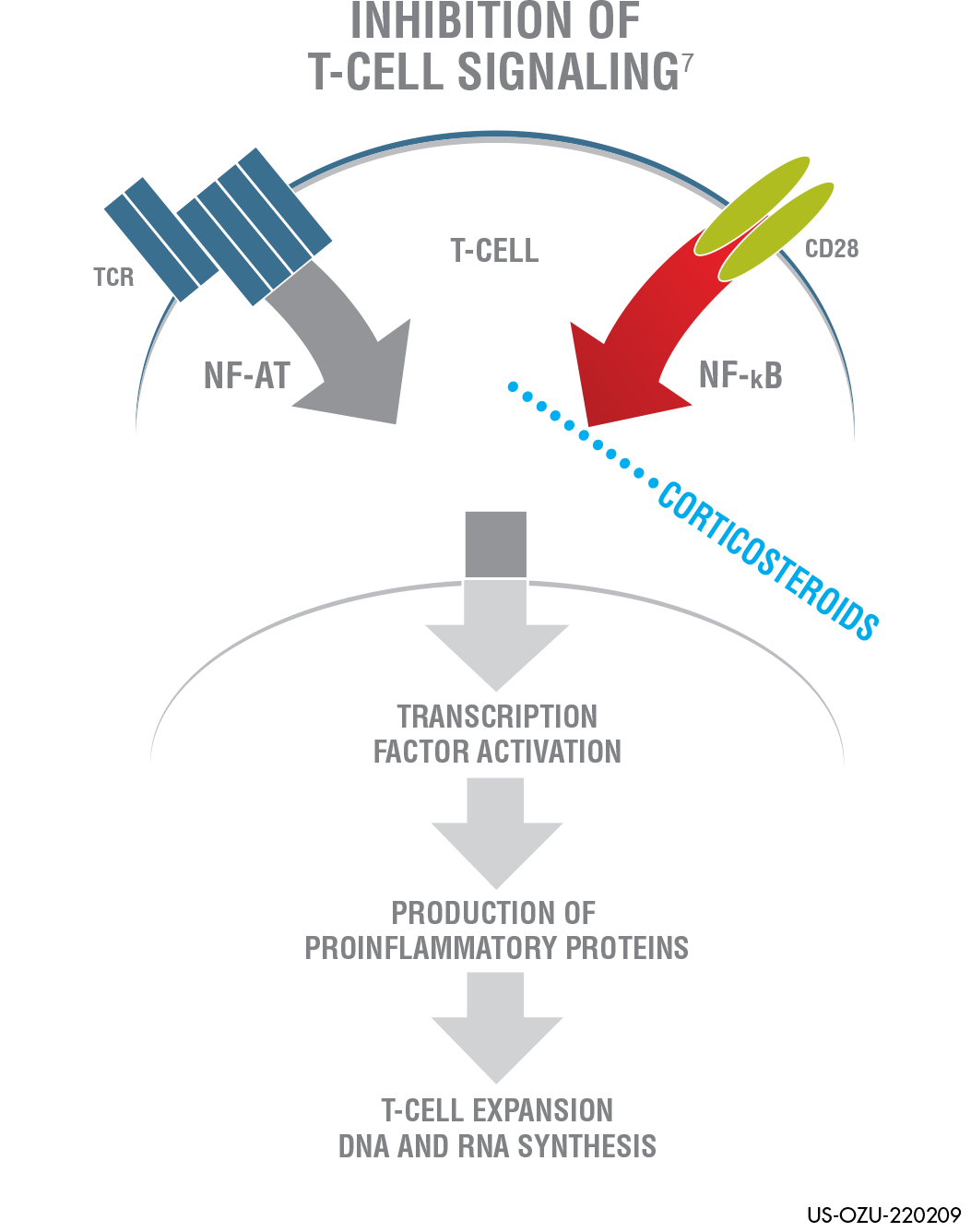
SEE HOW OZURDEX® WORKS TO TREAT
DIABETIC MACULAR EDEMA (DME)
SEE 3-YEAR CLINICAL EFFICACY
SEE HOW OZURDEX® WORKS TO TREAT
MACULAR EDEMA FOLLOWING RVO
(BRVO OR CRVO)
HOW OZURDEX® WORKS IN
NONINFECTIOUS POSTERIOR
SEGMENT UVEITIS
NOVADUR® TECHNOLOGY:
A PROPRIETARY DRUG DELIVERY SYSTEM

Tiny
implant

Before
implantation

3 weeks
after implantation
Scanning electron microscopy (SEM) magnification of implant surface in an animal model. Clinical significance unknown.3
A PATENTED
INTRAVITREAL APPLICATOR

SEE HOW THE OZURDEX®
APPLICATOR WORKS
Click here to access injection technique educational resources.
Indications and Usage
Diabetic Macular Edema
OZURDEX® (dexamethasone intravitreal implant) is a corticosteroid indicated for the treatment of diabetic macular edema.
Retinal Vein Occlusion
OZURDEX® is a corticosteroid indicated for the treatment of macular edema following branch retinal vein occlusion (BRVO) or central retinal vein occlusion (CRVO).
Posterior Segment Uveitis
OZURDEX® is indicated for the treatment of noninfectious uveitis affecting the posterior segment of the eye.
Dosage and Administration
FOR OPHTHALMIC INTRAVITREAL INJECTION. The intravitreal injection procedure should be carried out under controlled aseptic conditions. Following the intravitreal injection, patients should be monitored for elevation in intraocular pressure and for endophthalmitis. Patients should be instructed to report any symptoms suggestive of endophthalmitis without delay.
IMPORTANT SAFETY INFORMATION
Contraindications
Ocular or Periocular Infections: OZURDEX® (dexamethasone intravitreal implant) is contraindicated in patients with active or suspected ocular or periocular infections including most viral diseases of the cornea and conjunctiva, including active epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, varicella, mycobacterial infections, and fungal diseases.
Glaucoma: OZURDEX® is contraindicated in patients with glaucoma, who have cup to disc ratios of greater than 0.8.
Torn or Ruptured Posterior Lens Capsule: OZURDEX® is contraindicated in patients whose posterior lens capsule is torn or ruptured because of the risk of migration into the anterior chamber. Laser posterior capsulotomy in pseudophakic patients is not a contraindication for OZURDEX® use.
Hypersensitivity: OZURDEX® is contraindicated in patients with known hypersensitivity to any components of this product.
Warnings and Precautions
Intravitreal Injection-related Effects: Intravitreal injections, including those with OZURDEX®, have been associated with endophthalmitis, eye inflammation, increased intraocular pressure, and retinal detachments. Patients should be monitored regularly following the injection.
Steroid-related Effects: Use of corticosteroids including OZURDEX® may produce posterior subcapsular cataracts, increased intraocular pressure, glaucoma, and may enhance the establishment of secondary ocular infections due to bacteria, fungi, or viruses.
Corticosteroids are not recommended to be used in patients with a history of ocular herpes simplex because of the potential for reactivation of the viral infection.
Adverse Reactions
Diabetic Macular Edema
Ocular adverse reactions reported by greater than or equal to 1% of patients in the two combined 3-year clinical trials following injection of OZURDEX® for diabetic macular edema include: cataract (68%), conjunctival hemorrhage (23%), visual acuity reduced (9%), conjunctivitis (6%), vitreous floaters (5%), conjunctival edema (5%), dry eye (5%), vitreous detachment (4%), vitreous opacities (3%), retinal aneurysm (3%), foreign body sensation (2%), corneal erosion (2%), keratitis (2%), anterior chamber inflammation (2%), retinal tear (2%), eyelid ptosis (2%). Non-ocular adverse reactions reported by greater than or equal to 5% of patients include: hypertension (13%) and bronchitis (5%).
Increased Intraocular Pressure: IOP elevation greater than or equal to 10 mm Hg from baseline at any visit was seen in 28% of OZURDEX® patients versus 4% of sham patients. 42% of the patients who received OZURDEX® were subsequently treated with IOP-lowering medications during the study versus 10% of sham patients.
The increase in mean IOP was seen with each treatment cycle, and the mean IOP generally returned to baseline between treatment cycles (at the end of the 6-month period).
Cataracts and Cataract Surgery: The incidence of cataract development in patients who had a phakic study eye was higher in the OZURDEX® group (68%) compared with Sham (21%). The median time of cataract being reported as an adverse event was approximately 15 months in the OZURDEX® group and 12 months in the Sham group. Among these patients, 61% of OZURDEX® subjects versus 8% of sham-controlled subjects underwent cataract surgery, generally between Month 18 and Month 39 (Median Month 21 for OZURDEX® group and 20 for Sham) of the studies.
Retinal Vein Occlusion and Posterior Segment Uveitis
Adverse reactions reported by greater than 2% of patients in the first 6 months following injection of OZURDEX® for retinal vein occlusion and posterior segment uveitis include: intraocular pressure increased (25%), conjunctival hemorrhage (22%), eye pain (8%), conjunctival hyperemia (7%), ocular hypertension (5%), cataract (5%), vitreous detachment (2%), and headache (4%).
Increased IOP with OZURDEX® peaked at approximately week 8. During the initial treatment period, 1% (3/421) of the patients who received OZURDEX® required surgical procedures for management of elevated IOP.
Please see accompanying full Prescribing Information or visit https://www.rxabbvie.com/pdf/ozurdex_pi.pdf
US-OZU-230032